How To Draw Line Bond Structures
Molecular Shape |
|---|
The Shape of Molecules
The three dimensional shape or configuration of a molecule is an important characteristic. This shape is dependent on the preferred spatial orientation of covalent bonds to atoms having two or more bonding partners. Three dimensional configurations are best viewed with the aid of models.  In gild to stand for such configurations on a two-dimensional surface (newspaper, blackboard or screen), we often use perspective drawings in which the direction of a bond is specified by the line connecting the bonded atoms.
In gild to stand for such configurations on a two-dimensional surface (newspaper, blackboard or screen), we often use perspective drawings in which the direction of a bond is specified by the line connecting the bonded atoms. 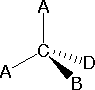 In about cases the focus of configuration is a carbon atom then the lines specifying bond directions will originate there. As defined in the diagram on the right, a uncomplicated direct line represents a bond lying approximately in the surface aeroplane. The 2 bonds to substituents A in the construction on the left are of this kind. A wedge shaped bond is directed in front of this airplane (thick end toward the viewer), as shown by the bail to substituent B; and a hatched bond is directed in back of the aeroplane (away from the viewer), every bit shown by the bond to substituent D. Some texts and other sources may utilize a dashed bond in the same manner as we have defined the hatched bond, just this can be confusing because the dashed bond is oftentimes used to represent a partial bond (i.east. a covalent bond that is partially formed or partially broken). The following examples make use of this note, and also illustrate the importance of including non-bonding valence beat electron pairs (colored blue) when viewing such configurations .
In about cases the focus of configuration is a carbon atom then the lines specifying bond directions will originate there. As defined in the diagram on the right, a uncomplicated direct line represents a bond lying approximately in the surface aeroplane. The 2 bonds to substituents A in the construction on the left are of this kind. A wedge shaped bond is directed in front of this airplane (thick end toward the viewer), as shown by the bail to substituent B; and a hatched bond is directed in back of the aeroplane (away from the viewer), every bit shown by the bond to substituent D. Some texts and other sources may utilize a dashed bond in the same manner as we have defined the hatched bond, just this can be confusing because the dashed bond is oftentimes used to represent a partial bond (i.east. a covalent bond that is partially formed or partially broken). The following examples make use of this note, and also illustrate the importance of including non-bonding valence beat electron pairs (colored blue) when viewing such configurations .
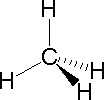 | 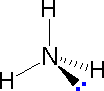 | 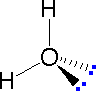 | ||
| Methane | Ammonia | Water |
|---|
Bonding configurations are readily predicted by valence-vanquish electron-pair repulsion theory, commonly referred to as VSEPR in most introductory chemical science texts. This simple model is based on the fact that electrons repel each other, and that it is reasonable to expect that the bonds and not-bonding valence electron pairs associated with a given atom volition prefer to be as far autonomously as possible. The bonding configurations of carbon are easy to recollect, since there are only 3 categories.
| Configuration | Bonding Partners | Bond Angles | Instance |
|---|---|---|---|
| Tetrahedral | four | 109.5º | |
| Trigonal | three | 120º | |
| Linear | ii | 180º |
In the three examples shown above, the key atom (carbon) does not have any not-bonding valence electrons; consequently the configuration may be estimated from the number of bonding partners alone. For molecules of h2o and ammonia, all the same, the non-bonding electrons must be included in the calculation. In each case there are four regions of electron density associated with the valence beat and so that a tetrahedral bond bending is expected. The measured bond angles of these compounds (H2O 104.5º & NHiii 107.3º) show that they are closer to being tetrahedral than trigonal or linear. Of form, it is the configuration of atoms (not electrons) that defines the the shape of a molecule, and in this sense ammonia is said to be pyramidal (not tetrahedral). The compound boron trifluoride, BF3, does not have not-bonding valence electrons and the configuration of its atoms is trigonal. Dainty treatments of VSEPR theory accept been provided by Oxford and Purdue . Click on the university name to visit their site.
The best fashion to study the three-dimensional shapes of molecules is by using molecular models. Many kinds of model kits are available to students and professional chemists. Some of the useful features of physical models can be approximated past the model viewing applet Jmol. This powerful visualization tool allows the user to move a molecular stucture in whatever way desired. Atom distances and angles are easily determined. To measure a distance, double-click on two atoms. To measure a bond angle, practise a double-click, single-click, double-click on three atoms. To measure a torsion angle, do a double-click, single-click, single-click, double-click on four atoms. A pop-upwardly menu of commands may be accessed past the correct button on a PC or a control-click on a Mac while the cursor is inside the display frame.
You may examine several Jmol models of compounds discussed above by .
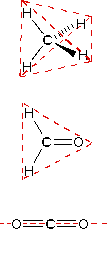
One style in which the shapes of molecules manifest themselves experimentally is through molecular dipole moments. A molecule which has one or more polar covalent bonds may accept a dipole moment as a event of the accumulated bond dipoles. In the instance of h2o, we know that the O-H covalent bond is polar, due to the different electronegativities of hydrogen and oxygen. Since there are two O-H bonds in water, their bond dipoles will interact and may result in a molecular dipole which can exist measured. The following diagram shows four possible orientations of the O-H bonds.
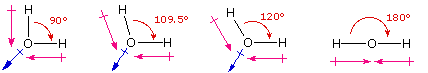
The bond dipoles are colored magenta and the resulting molecular dipole is colored bluish. In the linear configuration (bond angle 180º) the bond dipoles cancel, and the molecular dipole is nothing. For other bail angles (120 to 90º) the molecular dipole would vary in size, being largest for the 90º configuration. In a similar style the configurations of marsh gas (CH4) and carbon dioxide (COtwo) may be deduced from their nothing molecular dipole moments. Since the bond dipoles take canceled, the configurations of these molecules must be tetrahedral (or square-planar) and linear respectively.
The case of methane provides insight to other arguments that accept been used to confirm its tetrahedral configuration. For purposes of word we shall consider three other configurations for CHiv, square-planar, square-pyramidal and triangular-pyramidal.
Models of these possibilities may be examined by .
Substitution of 1 hydrogen by a chlorine atom gives a CHiiiCl compound. Since the tetrahedral, square-planar and foursquare-pyramidal configurations have structurally equivalent hydrogen atoms, they would each requite a single substitution production. Still, in the trigonal-pyramidal configuration one hydrogen (the noon) is structurally unlike from the other three (the pyramid base). Commutation in this example should give two different CHthreeCl compounds if all the hydrogens react. In the case of disubstitution, the tetrahedral configuration of methane would pb to a single CH2Clii product, but the other configurations would requite two unlike CH2Cl2 compounds. These substitution possibilities are shown in the above insert.
Isomers |
|---|
Isomers
Structural Formulas
It is necessary to draw structural formulas for organic compounds considering in virtually cases a molecular formula does non uniquely represent a single compound. Different compounds having the same molecular formula are called isomers, and the prevalence of organic isomers reflects the boggling versatility of carbon in forming potent bonds to itself and to other elements.
When the grouping of atoms that make up the molecules of dissimilar isomers are bonded together in fundamentally different ways, we refer to such compounds as ramble isomers. There are 7 constitutional isomers of CivHtenO, and structural formulas for these are drawn in the following table. These formulas represent all known and possible C4HxO compounds, and display a mutual structural feature. At that place are no double or triple bonds and no rings in whatever of these structures.. Note that each of the carbon atoms is bonded to four other atoms, and is saturated with bonding partners.
| Kekulé Formula | Condensed Formula | Shorthand Formula |
|---|---|---|
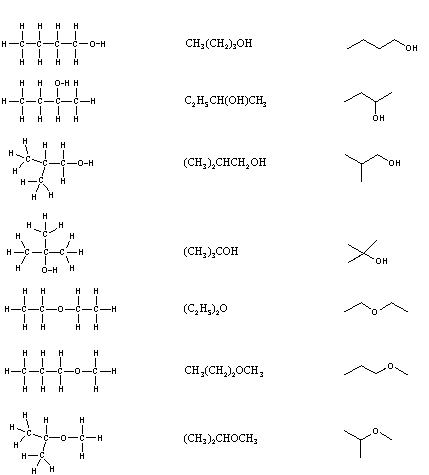 | ||
Simplification of structural formulas may exist achieved without any loss of the information they convey. In condensed structural formulas the bonds to each carbon are omitted, but each distinct structural unit (group) is written with subscript numbers designating multiple substituents, including the hydrogens. Shorthand (line) formulas omit the symbols for carbon and hydrogen entirely. Each straight line segment represents a bond, the ends and intersections of the lines are carbon atoms, and the correct number of hydrogens is calculated from the tetravalency of carbon. Not-bonding valence crush electrons are omitted in these formulas.
Developing the power to visualize a 3-dimensional structure from two-dimensional formulas requires practice, and in virtually cases the aid of molecular models. As noted earlier, many kinds of model kits are available to students and professional chemists, and the starting time student is encouraged to obtain i.
| Constitutional isomers have the same molecular formula, just their concrete and chemic properties may be very dissimilar. |
|---|
Distinguishing Carbon Atoms
When discussing structural formulas, it is oft useful to distinguish different groups of carbon atoms past their structural characteristics. A main carbon (1º) is one that is bonded to no more than 1 other carbon atom. A secondary carbon (2º) is bonded to 2 other carbon atoms, and tertiary (3º) and quaternary (4º) carbon atoms are bonded respectively to three and four other carbons. The 3 C5H12 isomers shown below illustrate these terms.
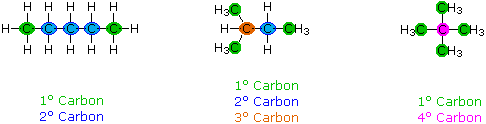
Structural differences may occur within these four groups, depending on the molecular constitution. In the formula on the correct all four 1º-carbons are structurally equivalent (call back the tetrahedral configuration of tetravalent carbon); nonetheless the central formula has 2 equivalent 1º-carbons (bonded to the 3º carbon on the left cease) and a unmarried, structurally different 1º-carbon (bonded to the 2º-carbon) at the right end. Similarly, the left-most formula has two structurally equivalent 2º-carbons (next to the ends of the chain), and a structurally different 2º-carbon in the middle of the chain. A consideration of molecular symmetry helps to distinguish structurally equivalent from nonequivalent atoms and groups. The ability to distinguish structural differences of this kind is an essential role of mastering organic chemistry. It volition come with practise and feel.
| Our ability to draw structural formulas for molecules is remarkable. |
|---|
Formula Analysis |
|---|
Analysis of Molecular Formulas
Although structural formulas are essential to the unique clarification of organic compounds, it is interesting and instructive to evaluate the information that may be obtained from a molecular formula lonely. 3 useful rules may exist listed:
- The number of hydrogen atoms that can be bonded to a given number of carbon atoms is limited by the valence of carbon. For compounds of carbon and hydrogen (hydrocarbons) the maximum number of hydrogen atoms that can be bonded to n carbons is 2n + two (n is an integer). In the instance of methane, CH4, north=1 & 2n + 2 = 4. The origin of this formula is evident past considering a hydrocarbon fabricated upwards of a chain of carbon atoms. Here the eye carbons will each accept two hydrogens and the two end carbons have three hydrogens each. Thus, a vi-carbon chain (n = 6) may be written H-(CH2)half dozen-H, and the full hydrogen count is (2 ten 6) + 2 = 14. The presence of oxygen (valence = two) does not modify this human relationship, so the previously described C4HxO isomers follow the rule, n=4 & 2n + 2 = x. Halogen atoms (valence = 1) should be counted equivalent to hydrogen, as illustrated by CiiiHfiveCl3, n = 3 & 2n + 2 = viii = (5 + 3). If nitrogen is present, each nitrogen atom (valence = iii) increases the maximum number of hydrogens by one.
Some Plausible
Molecular FormulasC7H16O3, C9H18, C15H28O3, CsixH16N2 Some Incommunicable
Molecular FormulasCeightH20Osix, C23H50, C5H10Cl4, CivH12NO
- For stable organic compounds the full number of odd-valenced atoms is even. Thus, when even-valenced atoms such as carbon and oxygen are bonded together in whatever number and in whatever manner, the number of remaining unoccupied bonding sites must exist fifty-fifty. If these sites are occupied by univalent atoms such as H, F, Cl, etc. their total number will necessarily be even. Nitrogen is also an odd-valenced cantlet (three), and if it occupies a bonding site on carbon it adds two additional bonding sites, thus maintaining the even/odd parity.
Some Plausible
Molecular FormulasCivHivCltwo, C5HixOBr, C5H11NO2, C12HeighteenNorth2FCl Some Impossible
Molecular FormulasC5H9O2, C4H5ClBr, C6H11N2O, CxH18NCltwo
- The number of hydrogen atoms in stable compounds of carbon, hydrogen & oxygen reflects the number of double bonds and rings in their structural formulas. Consider a hydrocarbon with a molecular structure consisting of a simple concatenation of four carbon atoms, CHiiiCHtwoCH2CHiii. The molecular formula is CfourHten (the maximum number of bonded hydrogens by the 2n + 2 rule). If the four carbon atoms form a band, two hydrogens must be lost. Similarly, the introduction of a double bond entails the loss of two hydrogens, and a triple bond the loss of iv hydrogens.
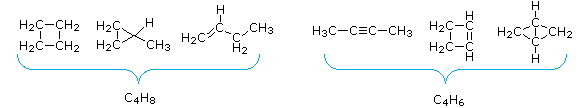
From the above discussion and examples it should be clear that the molecular formula of a hydrocarbon (CnHone thousand) provides information nigh the number of rings and/or double bonds that must be present in its structural formula. By rule #2 yard must be an fifty-fifty number, so if yard < (2n + 2) the difference is as well an even number that reflects any rings and double bonds. A triple bond is counted every bit ii double bonds.
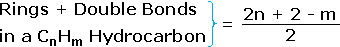
The presence of ane or more nitrogen atoms or element of group vii substituents requires a modified analysis. The above formula may be extended to such compounds by a few simple principles:
- The presence of oxygen does not modify the relationship.
- All halogens nowadays in the molecular formula must be replaced by hydrogen.
- Each nitrogen in the formula must be replaced by a CH moiety.
Resonance |
|---|
Resonance
Kekulé structural formulas are essential tools for understanding organic chemistry. However, the structures of some compounds and ions cannot be represented by a single formula. For example, sulfur dioxide (SO2) and nitric acid (HNO3) may each be described past 2 equivalent formulas (equations 1 & 2). For clarity the two ambiguous bonds to oxygen are given different colors in these formulas.
| one) sulfur dioxide | 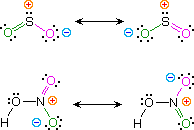 | |
| two) nitric acid |
If just i formula for sulfur dioxide was correct and accurate, and then the double bail to oxygen would be shorter and stronger than the single bail. Since experimental evidence indicates that this molecule is bent (bond angle 120º) and has equal length sulfur : oxygen bonds (i.432 Å), a single formula is inadequate, and the actual structure resembles an average of the ii formulas. This averaging of electron distribution over 2 or more than hypothetical contributing structures (canonical forms) to produce a hybrid electronic construction is chosen resonance. Likewise, the structure of nitric acid is best described as a resonance hybrid of 2 structures, the double headed arrow being the unique symbol for resonance.
The to a higher place examples stand for one farthermost in the application of resonance. Here, ii structurally and energetically equivalent electronic structures for a stable compound tin can be written, but no unmarried construction provides an accurate or even an adequate representation of the true molecule. In cases such as these, the electron delocalization described by resonance enhances the stability of the molecules, and compounds or ions equanimous of such molecules often evidence exceptional stability.
| 3) formaldehyde | 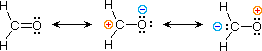 |
The electronic structures of most covalent compounds exercise not suffer the inadequacy noted in a higher place. Thus, completely satisfactory Kekulé formulas may be drawn for water (H2O), methane (CH4) and acetylene CiiH2). Nevertheless, the principles of resonance are very useful in rationalizing the chemical beliefs of many such compounds. For case, the carbonyl group of formaldehyde (the carbon-oxygen double bond) reacts readily to give improver products. The form of these reactions can be explained by a small contribution of a dipolar resonance contributor, as shown in equation 3. Hither, the first contributor (on the left) is clearly the best representation of this molecular unit, since there is no charge separation and both the carbon and oxygen atoms have achieved valence shell neon-like configurations by covalent electron sharing. If the double bond is broken heterolytically, formal accuse pairs effect, as shown in the other 2 structures. The preferred accuse distribution will have the positive charge on the less electronegative cantlet (carbon) and the negative charge on the more electronegative cantlet (oxygen). Therefore the centre formula represents a more reasonable and stable structure than the one on the correct. The application of resonance to this case requires a weighted averaging of these canonical structures. The double bonded structure is regarded as the major contributor, the centre structure a minor contributor and the right hand structure a non-contributor. Since the center, accuse-separated contributor has an electron deficient carbon atom, this explains the tendency of electron donors (nucleophiles) to bond at this site.
The basic principles of the resonance method may at present be summarized.
For a given chemical compound, a set of Lewis / Kekulé structures are written, keeping the relative positions of all the component atoms the aforementioned. These are the canonical forms to be considered, and all must have the same number of paired and unpaired electrons.
The following factors are important in evaluating the contribution each of these approved structures makes to the actual molecule.
- The number of covalent bonds in a structure. (The greater the bonding, the more important and stable the contributing structure.)
- Formal charge separation. (Other factors aside, charge separation decreases the stability and importance of the contributing construction.)
- Electronegativity of charge begetting atoms and charge density. (High accuse density is destabilizing. Positive accuse is best accommodated on atoms of depression electronegativity, and negative accuse on loftier electronegative atoms.)
The stability of a resonance hybrid is always greater than the stability of any canonical correspondent. Consequently, if one approved course has a much greater stability than all others, the hybrid will closely resemble information technology electronically and energetically. This is the case for the carbonyl group (eq.three). The left hand C=O construction has much greater total bonding than either accuse-separated construction, and then it describes this functional group rather well. On the other hand, if two or more canonical forms take identical depression energy structures, the resonance hybrid volition have exceptional stabilization and unique properties. This is the case for sulfur dioxide (eq.i) and nitric acid (eq.two).
| four) carbon monoxide | 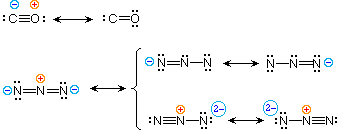 | |
| 5) azide anion |
To illustrate these principles we shall consider carbon monoxide (eq.4) and azide anion (eq.five). In each case the near stable approved form is on the left. For carbon monoxide, the additional bonding is more than important than charge separation. Furthermore, the double bonded structure has an electron deficient carbon atom (valence shell sextet). A similar destabilizing factor is present in the ii azide canonical forms on the pinnacle row of the bracket (three bonds vs. 4 bonds in the left nigh structure). The lesser row pair of structures have four bonds, but are destabilized by the high charge density on a single nitrogen atom.
All the examples on this page demonstrate an important restriction that must exist remembered when using resonance. No atoms change their positions within the common structural framework. Only electrons are moved.
Orbitals |
|---|
Atomic and Molecular Orbitals
A more than detailed model of covalent bonding requires a consideration of valence shell atomic orbitals. For second period elements such equally carbon, nitrogen and oxygen, these orbitals have been designated 2s, 2px, 2py & 2pz. The spatial distribution of electrons occupying each of these orbitals is shown in the diagram below.
Very nice displays of orbitals may exist institute at the following sites:
J. Gutow, Univ. Wisconsin Oshkosh
R. Spinney, Ohio State
M. Winter, Sheffield Academy
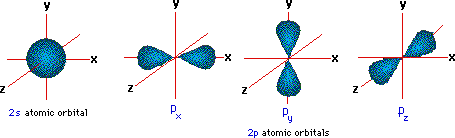
The valence trounce electron configuration of carbon is twosouthward 2, 2px 1, 2py 1 & 2pz 0. If this were the configuration used in covalent bonding, carbon would only exist able to form 2 bonds. In this case, the valence shell would accept six electrons- two shy of an octet. However, the tetrahedral structures of methane and carbon tetrachloride demonstrate that carbon tin form four equivalent bonds, leading to the desired octet. In order to explain this covalent bonding, Linus Pauling proposed an orbital hybridization model in which all the valence crush electrons of carbon are reorganized.
Hybrid Orbitals
In order to explicate the construction of methane (CH4), the 2s and three 2p orbitals are converted to 4 equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp3. These hybrid orbitals have a specific orientation, and the 4 are naturally oriented in a tetrahedral style. Thus, the four covalent bonds of methane consist of shared electron pairs with four hydrogen atoms in a tetrahedral configuration, every bit predicted by VSEPR theory.

Molecular Orbitals
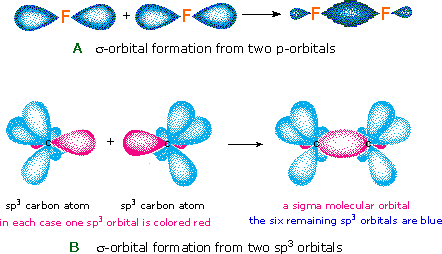
Just as the valence electrons of atoms occupy atomic orbitals (AO), the shared electron pairs of covalently bonded atoms may be thought of as occupying molecular orbitals (MO). It is convenient to estimate molecular orbitals by combining or mixing two or more atomic orbitals. In general, this mixing of n diminutive orbitals e'er generates north molecular orbitals. The hydrogen molecule provides a simple example of MO formation. In the post-obit diagram, two 1s atomic orbitals combine to give a sigma (σ) bonding (low free energy) molecular orbital and a 2d higher energy MO referred to as an antibonding orbital. The bonding MO is occupied past two electrons of contrary spin, the result being a covalent bond.

The notation used for molecular orbitals parallels that used for atomic orbitals. Thus, s-orbitals have a spherical symmetry surrounding a unmarried nucleus, whereas σ-orbitals have a cylindrical symmetry and encompass 2 (or more than) nuclei. In the instance of bonds between 2d period elements, p-orbitals or hybrid atomic orbitals having p-orbital character are used to form molecular orbitals. For example, the sigma molecular orbital that serves to bond two fluorine atoms together is generated by the overlap of p-orbitals (part A below), and two sp3 hybrid orbitals of carbon may combine to give a similar sigma orbital. When these bonding orbitals are occupied by a pair of electrons, a covalent bond, the sigma bond results. Although we have ignored the remaining p-orbitals, their inclusion in a molecular orbital treatment does not lead to whatever additional bonding, as may be shown by activating the fluorine correlation diagram below.
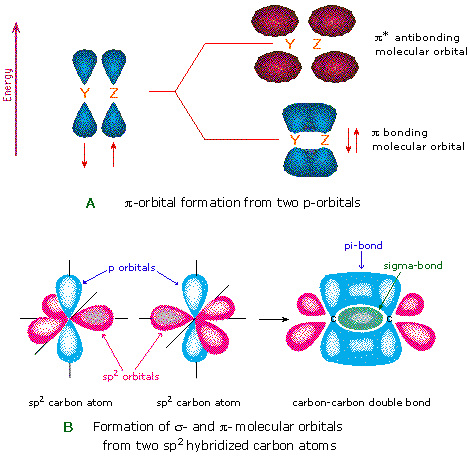
Another type of MO (the π orbital) may exist formed from 2 p-orbitals by a lateral overlap, as shown in part A of the following diagram. Since bonds consisting of occupied π-orbitals (pi-bonds) are weaker than sigma bonds, pi-bonding between 2 atoms occurs only when a sigma bond has already been established. Thus, pi-bonding is generally found only as a component of double and triple covalent bonds. Since carbon atoms involved in double bonds have only three bonding partners, they require only iii hybrid orbitals to contribute to three sigma bonds. A mixing of the 2s-orbital with 2 of the 2p orbitals gives three sp2 hybrid orbitals, leaving i of the p-orbitals unused. 2 sptwo hybridized carbon atoms are then joined together by sigma and pi-bonds (a double bail), every bit shown in function B.
 | |
|---|---|
The manner in which diminutive orbitals overlap to form molecular orbitals is actually more complex than the localized examples given above. These are useful models for explaining the structure and reactivity of many organic compounds, but modern molecular orbital theory involves the creation of an orbital correlation diagram. 2 examples of such diagrams for the simple diatomic elements Ftwo and N2 volition be fatigued above when the appropriate button is clicked. The 1s and 2s diminutive orbitals do not provide any overall bonding, since orbital overlap is minimal, and the resulting sigma bonding and antibonding components would cancel. In both these cases three 2p atomic orbitals combine to course a sigma and two pi-molecular orbitals, each as a bonding and antibonding pair. The overall bonding order depends on the number of antibonding orbitals that are occupied. The subtle change in the energy of the σ2p bonding orbital, relative to the two degenerate π-bonding orbitals, is due to s-p hybridization that is unimportant to the present give-and-take.
One instance of the reward offered by the molecular orbital approach to bonding is the oxygen molecule. Hither, the correlation diagram correctly accounts for the paramagnetic character of this simple diatomic chemical compound. Likewise, the orbital correlation diagram for methane provides some other example of the difference in electron density predicted by molecular orbital calculations from that of the localized bail model. Click on the compound names for these displays.
A cartoon of the p and π orbitals of a double bail may be examined past .
A model of the π orbitals of ethene may be examined by .
The p-orbitals in these model are represented by cherry-red and blue colored spheres or ellipses, which represent different phases, divers by the mathematical wave equations for such orbitals.
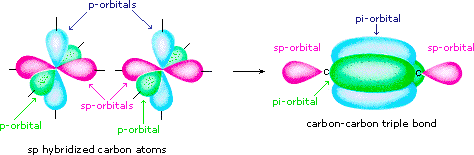
Finally, in the case of carbon atoms with only two bonding partners simply ii hybrid orbitals are needed for the sigma bonds, and these sp hybrid orbitals are directed 180º from each other. Two p-orbitals remain unused on each sp hybridized cantlet, and these overlap to give 2 pi-bonds following the germination of a sigma bond (a triple bail), as shown below.
The various hybridization states of carbon may exist examined by .
Do Problems |
|---|
The following problems explore many of the concepts discussed above. They include recognition of different molecular shapes, obtaining structural data from molecular formulas, evaluation of line and condensed structural formulas, formal charges, hybridization and resonance.
| Return to Table of Contents |
|---|
This page is the property of William Reusch. Comments, questions and errors should be sent to whreusch@msu.edu.
These pages are provided to the IOCD to assist in capacity edifice in chemic education. 05/05/2013
![]()
Source: https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/intro3.htm
Posted by: bealsgrany1997.blogspot.com


0 Response to "How To Draw Line Bond Structures"
Post a Comment